ఇటీవలి సంవత్సరాలలో, ఆరోగ్య సంరక్షణ వ్యక్తిగతీకరించిన వైద్యం వైపు అద్భుతమైన మార్పును చూసింది-రోగాలకు కాకుండా వ్యక్తికి చికిత్సను అందించే విధానం. ఈ మార్పుకు ప్రధాన కారణం ఫార్మాకోజెనోమిక్స్ (pharmacogenomics). ఒక వ్యక్తి యొక్క జన్యుపరమైన లక్షణాలు ఔషధాలకు ఎలా స్పందిస్తాయో ఇది అధ్యయనం చేస్తుంది. ఈ అత్యాధునిక రంగం మందులను సూచించే విధానంలో విప్లవాత్మక మార్పులు తీసుకురాగల సామర్థ్యాన్ని కలిగి ఉంది, దుష్ప్రభావాలను తగ్గించి, చికిత్సలను మరింత ప్రభావవంతంగా చేస్తుంది.
ఫార్మాకోజెనోమిక్స్ అంటే ఏమిటి?
ఫార్మాకోజెనోమిక్స్ ఫార్మకాలజీ (ఔషధాల శాస్త్రం) మరియు జెనోమిక్స్ (జన్యువులు మరియు వాటి విధుల అధ్యయనం) సూత్రాలను మిళితం చేస్తుంది. ముఖ్యంగా, ఇది మీ జన్యువులలోని వైవిధ్యాలు మీరు మందులను ఎలా జీవక్రియ చేస్తాయో ఎలా ప్రభావితం చేస్తాయో అర్థం చేసుకోవడమే దీని లక్ష్యం. వేర్వేరు వ్యక్తులకు ఒకే పరిస్థితి ఉండవచ్చు, కానీ ఒక నిర్దిష్ట ఔషధానికి వారు స్పందించే విధానం వారి జన్యు ప్రొఫైల్ కారణంగా చాలా భిన్నంగా ఉంటుంది.
ఉదాహరణకు, ఒక వ్యక్తికి సంపూర్ణంగా పనిచేసే ఔషధం మరొకరిపై తక్కువ ప్రభావాన్ని చూపవచ్చు లేదా అది తీవ్రమైన దుష్ప్రభావాలను కలిగించవచ్చు. ఈ ప్రతిస్పందనలను అంచనా వేయడానికి ఫార్మాకోజెనోమిక్స్ సహాయపడుతుంది, ప్రతి వ్యక్తికి అత్యంత ప్రభావవంతమైన మరియు సురక్షితమైన చికిత్సా ఎంపికలను ఎంచుకోవడానికి ఆరోగ్య సంరక్షణ ప్రదాతలను అనుమతిస్తుంది.
ఫార్మాకోజెనోమిక్స్ ఎందుకు ముఖ్యమైనది?
సాంప్రదాయ వైద్యంలో, చాలా చికిత్సలు "వన్-సైజ్-ఫిట్స్-ఆల్" విధానాన్ని అనుసరిస్తాయి. చాలా మందికి ఇది ప్రభావవంతంగా ఉన్నప్పటికీ, ఈ పద్ధతి తరచుగా ఔషధాల ప్రభావం మరియు భద్రతను ప్రభావితం చేసే ప్రత్యేకమైన జన్యు కారకాలను విస్మరిస్తుంది. వ్యక్తిగతీకరించిన వైద్యంలో ఫార్మాకోజెనోమిక్స్ ఎందుకు ముఖ్యమో ఇక్కడ వివరించబడింది:
-
ప్రత్యేకమైన ఔషధ ఎంపిక: రోగి DNAని విశ్లేషించడం ద్వారా, ఆరోగ్య సంరక్షణ ప్రదాతలు ఏ మందులు అత్యంత ప్రభావవంతంగా ఉంటాయో నిర్ణయించగలరు. ఇది చికిత్సలను సూచించడంలో తరచుగా ఎదురయ్యే అంచనాను తొలగిస్తుంది మరియు వేగవంతమైన, మరింత ఖచ్చితమైన ఫలితాలను నిర్ధారిస్తుంది.
-
ప్రతికూల ఔషధ ప్రతిచర్యల తగ్గింపు: ఫార్మాకోజెనోమిక్స్ యొక్క ముఖ్యమైన ప్రయోజనాలలో ఒకటి హానికరమైన దుష్ప్రభావాలను అంచనా వేయగల మరియు నిరోధించగల సామర్థ్యం. నిర్దిష్ట జన్యు వైవిధ్యాలు ఉన్న రోగులు మందులను చాలా వేగంగా లేదా చాలా నెమ్మదిగా జీవక్రియ చేయవచ్చు, ఇది రక్తప్రవాహంలో విషపూరిత స్థాయిలకు లేదా సరిపోని చికిత్సా ప్రభావాలకు దారితీస్తుంది. ఫార్మాకోజెనోమిక్స్ పరీక్ష చికిత్స ప్రారంభం కావడానికి ముందు అటువంటి ప్రమాదాలను గుర్తించగలదు.
-
మెరుగైన ఔషధ మోతాదు: సరైన మోతాదును కనుగొనడం తరచుగా ట్రయల్-అండ్-ఎర్రర్ ప్రక్రియ. ఫార్మాకోజెనోమిక్స్ ఒక వ్యక్తి యొక్క జన్యు ప్రొఫైల్ ఆధారంగా సరైన మోతాదును సూచించడానికి ఆరోగ్య సంరక్షణ ప్రదాతలను అనుమతిస్తుంది, తక్కువ దుష్ప్రభావాలతో గరిష్ట ప్రభావాన్ని నిర్ధారిస్తుంది.
-
వ్యక్తిగతీకరించిన చికిత్సా ప్రణాళికలు: ఫార్మాకోజెనోమిక్స్ పరీక్ష నుండి వచ్చిన సమాచారంతో, ఆరోగ్య సంరక్షణ ప్రదాతలు అత్యంత వ్యక్తిగతీకరించిన చికిత్సా ప్రణాళికలను రూపొందించగలరు. ఇది రోగి ఫలితాలను మెరుగుపరుస్తుంది మరియు అనేక మందులను ప్రయత్నించడంలో గడిపిన సమయాన్ని తగ్గిస్తుంది.
ఫార్మాకోజెనోమిక్స్ ఎలా పనిచేస్తుంది?
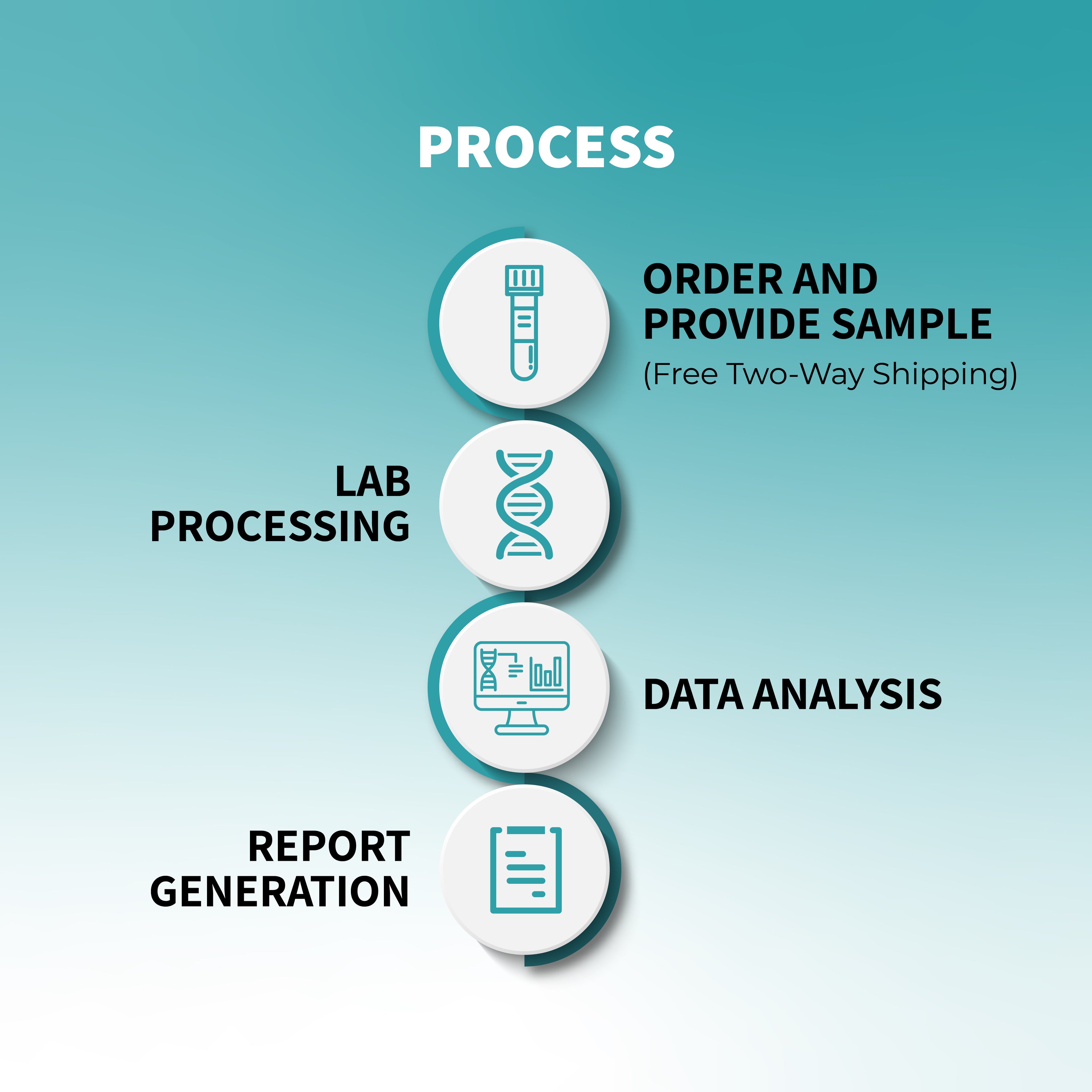
ఫార్మాకోజెనోమిక్ పరీక్ష సాధారణంగా సాధారణ లాలాజలం లేదా రక్త నమూనాతో జరుగుతుంది. నమూనాలోని DNA నిర్దిష్ట జన్యు వైవిధ్యాల కోసం విశ్లేషించబడుతుంది, ఇవి ఔషధ జీవక్రియను ప్రభావితం చేస్తాయి. పరీక్ష ఫలితాలు రోగికి ఏ మందులు ఉత్తమంగా పనిచేస్తాయో మరియు ఏవి ప్రతికూల ప్రభావాలను కలిగించవచ్చో నిర్ణయించడంలో సహాయపడతాయి.
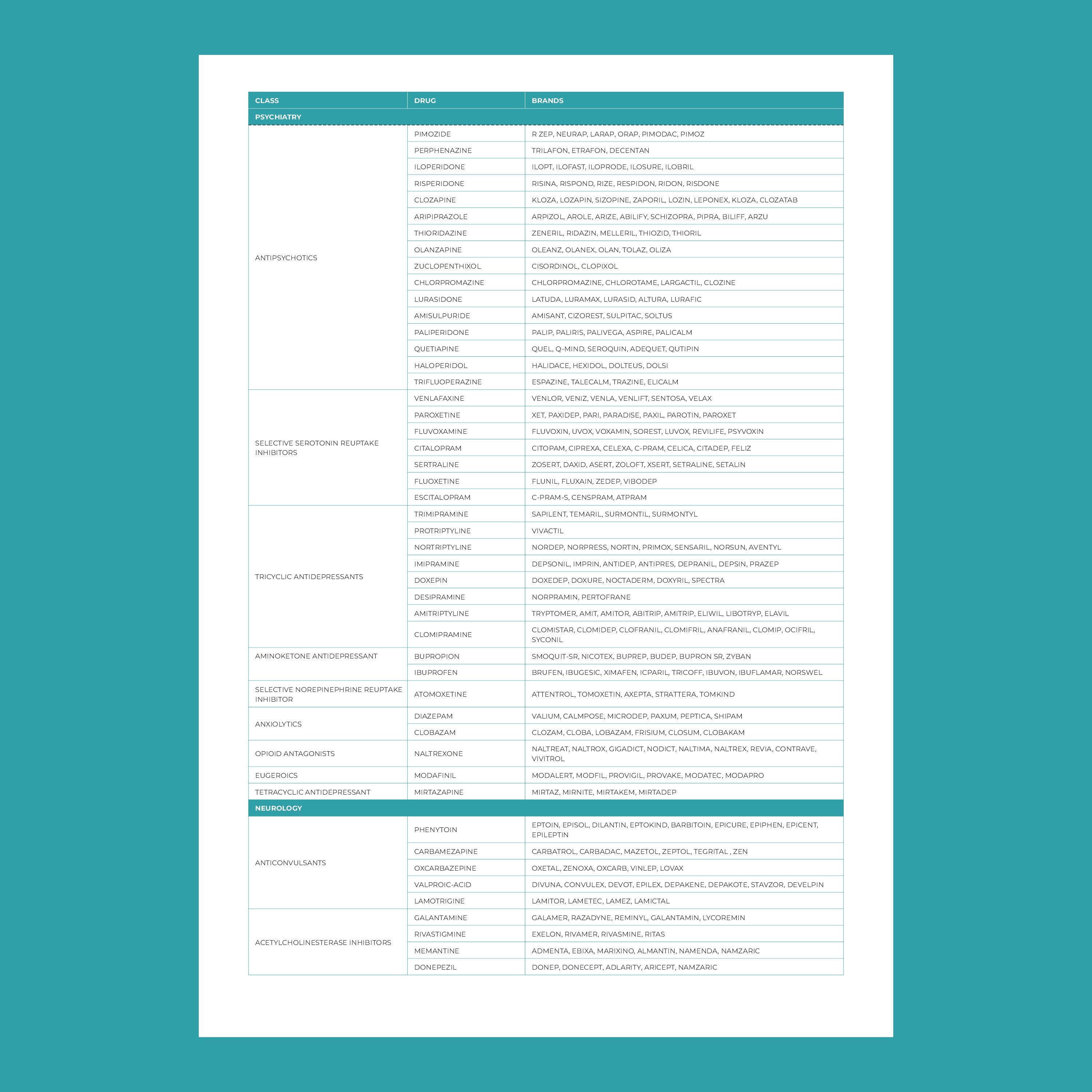
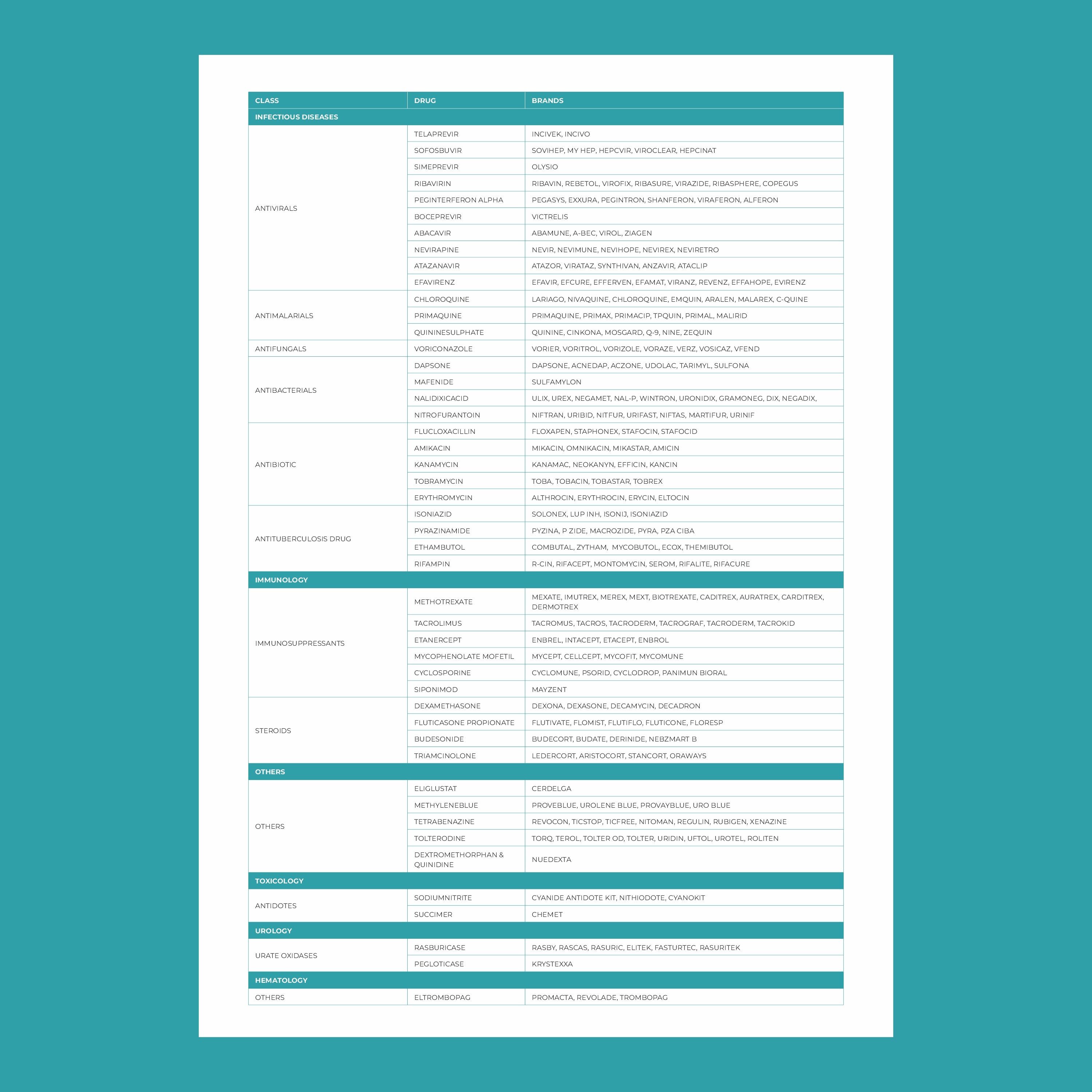
ఫార్మాకోజెనోమిక్స్లో కీలకమైన జన్యువులు:
- CYP450 ఎంజైమ్లు: ఇవి యాంటీడిప్రెసెంట్స్, యాంటిసైకోటిక్స్ మరియు నొప్పి నివారణలతో సహా అనేక మందులను జీవక్రియ చేయడానికి బాధ్యత వహిస్తాయి. ఈ జన్యువులలోని వైవిధ్యాలు ఒక ఔషధం ఎంత వేగంగా లేదా నెమ్మదిగా జీవక్రియ చేయబడుతుందో ప్రభావితం చేస్తాయి.
- VKORC1: ఈ జన్యువు వార్ఫరిన్, ఒక సాధారణ రక్తస్రావం నివారణ ఔషధం, ప్రతిస్పందనను ప్రభావితం చేస్తుంది. VKORC1 యొక్క నిర్దిష్ట వైవిధ్యాలు ఉన్న వ్యక్తులకు అధిక రక్తస్రావాన్ని నివారించడానికి తక్కువ మోతాదు అవసరం కావచ్చు.
- SLCO1B1: ఈ జన్యువులోని వైవిధ్యాలు స్టాటిన్లు (కొలెస్ట్రాల్ తగ్గించడానికి ఉపయోగించేవి) శరీరంలో ఎలా ప్రాసెస్ చేయబడతాయో ప్రభావితం చేయవచ్చు. కొంతమంది వ్యక్తులు కండరాల నొప్పి వంటి దుష్ప్రభావాలకు ఎక్కువగా గురయ్యే అవకాశం ఉంది.
ఆచరణలో ఫార్మాకోజెనోమిక్స్: కేస్ స్టడీస్
వ్యక్తిగతీకరించిన వైద్యంలో ఫార్మాకోజెనోమిక్స్ ప్రభావాన్ని బాగా అర్థం చేసుకోవడానికి, కొన్ని ఉదాహరణలను చూద్దాం:
1. మానసిక ఆరోగ్య మందులు
మానసిక కుంగుబాటు లేదా ఆందోళన ఉన్న చాలా మంది రోగులు సరైన యాంటీడిప్రెసెంట్ను కనుగొనడానికి సుదీర్ఘ ట్రయల్ అండ్ ఎర్రర్ ప్రక్రియ ద్వారా వెళ్తారు. ఫార్మాకోజెనోమిక్స్ ఈ ప్రక్రియను క్రమబద్ధీకరించగలదు. ఔషధ జీవక్రియలో పాల్గొన్న జన్యువులను విశ్లేషించడం ద్వారా, ఆరోగ్య సంరక్షణ ప్రదాతలు ఏ యాంటీడిప్రెసెంట్లు ఎక్కువగా పనిచేస్తాయో మరియు ఏవి దుష్ప్రభావాలను కలిగించవచ్చో అంచనా వేయగలరు. ఉదాహరణకు, CYP2C19 జన్యువులోని వైవిధ్యాలు SSRIల (యాంటీడిప్రెసెంట్ యొక్క సాధారణ రకం) ప్రాసెసింగ్ను ప్రభావితం చేస్తాయి, ప్రభావం మరియు దుష్ప్రభావాల ప్రమాదాన్ని ప్రభావితం చేస్తాయి.
2. గుండె జబ్బులు
గుండె సంబంధిత వ్యాధులను నిర్వహించడంలో ఫార్మాకోజెనోమిక్స్ కీలక పాత్ర పోషిస్తుంది. ఉదాహరణకు, క్లోపిడోగ్రెల్ (స్ట్రోకులు మరియు గుండెపోటులను నివారించడానికి ఉపయోగించే ఔషధం) కాలేయంలోని ఎంజైమ్ల ద్వారా సక్రియం చేయబడాలి. CYP2C19 జన్యువులో నిర్దిష్ట జన్యు వైవిధ్యాలు ఉన్న వ్యక్తులు ఔషధాన్ని సమర్థవంతంగా సక్రియం చేయలేకపోవచ్చు, దీనివల్ల అది తక్కువ ప్రభావవంతంగా ఉంటుంది. జన్యు పరీక్ష రోగులకు సరైన యాంటీప్లేట్లెట్ చికిత్సను పొందేలా చూస్తుంది, గుండెపోటు లేదా స్ట్రోకుల ప్రమాదాన్ని తగ్గిస్తుంది.
3. క్యాన్సర్ చికిత్స
క్యాన్సర్ చికిత్స వ్యక్తిగతీకరించిన వైద్యం, ముఖ్యంగా ఫార్మాకోజెనోమిక్స్ కోసం ఒక ప్రముఖ ప్రాంతం. టామక్సిఫెన్ (రొమ్ము క్యాన్సర్ కోసం ఉపయోగించేది) వంటి మందులు కాలేయంలోని ఎంజైమ్ల ద్వారా సక్రియం చేయబడాలి. CYP2D6 జన్యువులోని జన్యు వైవిధ్యాలు ఔషధం ఎంత బాగా పనిచేస్తుందో ప్రభావితం చేయగలవు. ఫార్మాకోజెనోమిక్ పరీక్ష ఆంకాలజిస్టులకు చికిత్స యొక్క ఉత్తమ మార్గాన్ని నిర్ణయించడంలో సహాయపడుతుంది, క్యాన్సర్ ఉన్న రోగుల ఫలితాలను మెరుగుపరుస్తుంది.
ఫార్మాకోజెనోమిక్స్ యొక్క భవిష్యత్తు
ఫార్మాకోజెనోమిక్స్ రంగం వేగంగా అభివృద్ధి చెందుతోంది, కొత్త ఆవిష్కరణలు నిరంతరం జరుగుతున్నాయి. జన్యు పరీక్ష మరింత సరసమైనది మరియు అందుబాటులో ఉన్నందున, వైద్యంలోని అనేక రంగాలలో ఫార్మాకోజెనోమిక్-మార్గదర్శక చికిత్స వైపు గణనీయమైన మార్పును మనం ఆశించవచ్చు.
క్షితిజంపై ఉన్న కొన్ని ఉత్తేజకరమైన పరిణామాలు:
- విస్తృతమైన పరీక్ష: ఫార్మాకోజెనోమిక్స్ పరీక్ష వైద్య సంరక్షణలో ఒక సాధారణ భాగంగా మారవచ్చు. రక్త పరీక్షల వలె, ఔషధ జీవక్రియ కోసం జన్యు పరీక్ష చికిత్స ప్రారంభంలోనే చేయబడవచ్చు, చికిత్సను ప్రారంభం నుండి మార్గనిర్దేశం చేయడానికి.
- ఆర్టిఫిషియల్ ఇంటెలిజెన్స్ (AI) అనుసంధానం: AI శక్తితో, ఫార్మాకోజెనోమిక్స్ డేటాను త్వరగా ప్రాసెస్ చేయవచ్చు మరియు విశ్లేషించవచ్చు, రోగుల జన్యు ప్రొఫైల్స్ ఆధారంగా ఉత్తమ చికిత్సా ఎంపికల గురించి తక్షణ అంతర్దృష్టులను అందిస్తుంది.
- ఓవర్-ది-కౌంటర్ మందులకు విస్తరణ: ఫార్మాకోజెనోమిక్స్ ప్రిస్క్రిప్షన్ డ్రగ్స్ దాటి ఓవర్-ది-కౌంటర్ మందులకు విస్తరించగలదు. ఉదాహరణకు, కొంతమందికి ఐబుప్రోఫెన్ వంటి సాధారణ నొప్పి నివారణల నుండి దుష్ప్రభావాలకు జన్యుపరమైన ప్రవృత్తి ఉండవచ్చు.
ఫార్మాకోజెనోమిక్స్ లోని సవాళ్లు
ఫార్మాకోజెనోమిక్స్ యొక్క సామర్థ్యం అపారమైనది అయినప్పటికీ, దాని విస్తృతమైన స్వీకరణకు సవాళ్లు ఉన్నాయి:
-
ఖర్చు మరియు అందుబాటు: జన్యు పరీక్షల ఖర్చు తగ్గినప్పటికీ, చాలా మందికి ఇది ఇప్పటికీ ఒక అడ్డంకిగా ఉంటుంది. విస్తృతమైన స్వీకరణ పరీక్షను మరింత సరసమైనదిగా చేయడం మరియు బీమా కవరేజీని నిర్ధారించడంపై ఆధారపడి ఉంటుంది.
-
జ్ఞాన అంతరం: చాలా మంది ఆరోగ్య సంరక్షణ ప్రదాతలు ఇంకా ఫార్మాకోజెనోమిక్స్లో లేదా పరీక్ష ఫలితాలను ఎలా అర్థం చేసుకోవాలో పూర్తిగా శిక్షణ పొందకపోవచ్చు. ఫార్మాకోజెనోమిక్స్ను సాధారణ సంరక్షణలో అనుసంధానించడానికి కొనసాగుతున్న విద్య అవసరం.
-
నైతిక పరిశీలనలు: ఏదైనా జన్యు పరీక్ష వలె, గోప్యత మరియు జన్యు సమాచారం ఎలా ఉపయోగించబడవచ్చు అనే దాని గురించి ఆందోళనలు ఉన్నాయి. ఫార్మాకోజెనోమిక్స్ మరింత సాధారణం అయినందున స్పష్టమైన నిబంధనలు మరియు రక్షణలు కీలకం అవుతాయి.
ముగింపు
ఫార్మాకోజెనోమిక్స్ వ్యక్తిగతీకరించిన వైద్యానికి ముందంజలో ఉంది, ప్రతి వ్యక్తి యొక్క జన్యు ప్రొఫైల్కు అనుగుణంగా చికిత్సలు అందించే భవిష్యత్తును అందిస్తుంది. ప్రతికూల ఔషధ ప్రతిచర్యలను తగ్గించడం, ఔషధ ప్రభావాన్ని మెరుగుపరచడం మరియు చికిత్సా ప్రణాళికలను వ్యక్తిగతీకరించడం ద్వారా, ఫార్మాకోజెనోమిక్స్ ఆరోగ్య సంరక్షణను మార్చగల సామర్థ్యాన్ని కలిగి ఉంది. ఈ రంగం అభివృద్ధి చెందుతూనే ఉన్నందున, రోగులు మరియు ఆరోగ్య సంరక్షణ ప్రదాతలు ఇద్దరూ ఇది అందించే ఖచ్చితత్వం మరియు అంతర్దృష్టి నుండి ప్రయోజనం పొందుతారు.